ISO 13485 · Medical Manufacturing
PRECISION LIFELINES
In the world of medical device manufacturing, precision and reliability aren’t just goals—they’re lifelines. From intricate surgical tools to patient-specific implants, the components demand methods that deliver consistency, biocompatibility, and intricate detail. Enter medical device vacuum casting: a versatile, cost-effective process that excels at producing high-quality, small-to-medium batch parts for critical medical applications.
At Goldcattle, with decades of expertise and ISO 13485 certification, we’ve honed vacuum casting to meet the strictest medical standards. This article explores how it works, why it’s ideal for medical devices, and how we deliver excellence in every cast.
⚙️ VACUUM CASTING PRINCIPLE
vacuum
tolerance
materials
Figure 1: Vacuum casting chamber with medical-grade silicone mold. Air bubbles eliminated under vacuum ensures uniform density.
Advanced Vacuum Casting Process for Medical Devices
Our medical-grade vacuum casting process involves precise control at every stage to ensure the highest quality components. The process begins with creating a master model using CNC machining or 3D printing, which is then used to produce a silicone mold. Under vacuum conditions, liquid resin is drawn into the mold cavity, eliminating air bubbles and ensuring complete filling of even the most intricate features.
💡 Technical advantage: By maintaining vacuum pressure ≤5 mbar throughout the casting process, we achieve bubble-free parts with uniform density and surface finish. This is critical for medical devices where even microscopic defects can compromise performance or biocompatibility.
Key Process Parameters
- Vacuum pressure: ≤5 mbar during casting
- Resin temperature: 40-60°C for optimal flow
- Curing time: 1-4 hours depending on material
- Mold temperature: 60-80°C for consistent curing

Figure 2: Advanced vacuum casting equipment showing the complete process from mold preparation to finished part production.
Medical-Grade Materials for Vacuum Casting

Figure 3: Comprehensive display of medical-grade vacuum casting materials with technical specifications.
Material Selection Guide
🔬 Silicone Rubber (ISO 10993 Compliant)
– Shore hardness: 10A to 80A
– Biocompatible and hypoallergenic
– Temperature resistance: -60°C to 200°C
– Applications: Wound care products, catheter tips, prosthetic liners
💎 Polyurethane Resins
– Tensile strength: Up to 60 MPa
– Impact resistance: Excellent
– Sterilization compatible: Autoclave, EtO, gamma
– Applications: Orthopedic braces, surgical instrument handles
🔧 ABS-Like Resins
– High dimensional stability
– Good chemical resistance
– Surface finish: Smooth, paintable
– Applications: Diagnostic device enclosures, infusion pump housings
Key Benefits of Vacuum Casting for Medical Applications
🧬
1. Biocompatible Material Versatility
Medical-grade silicone (ISO 10993 compliant), flexible & hypoallergenic – wound care, catheter tips. Polyurethane resins: tensile up to 60 MPa for orthopaedic braces. ABS-like resins for enclosures.
🔬
2. Precision for Tight Tolerances
Achieve tolerances as tight as ±0.1mm for parts under 100mm, with consistent repeatability. For a portable ultrasound probe housing, we maintained ±0.05mm on critical mating surfaces, ensuring waterproof seal after autoclaving (134°C, 3 bar).
💰
3. Cost-Effective Small-to-Medium Batches
Silicone molds $500–$2,000 vs. steel tooling. Produce 20–500 parts per mold. Goldcattle reduced a client’s prototyping costs by 40% (infusion pump components, 50 functional prototypes in 7 days).
⏱️
4. Rapid Turnaround
Silicone mold in 3–5 days, part production 1–3 days per batch. Emergency ventilator valve components: 200 parts delivered in 48h for clinical trial.
Goldcattle’s Customization Process
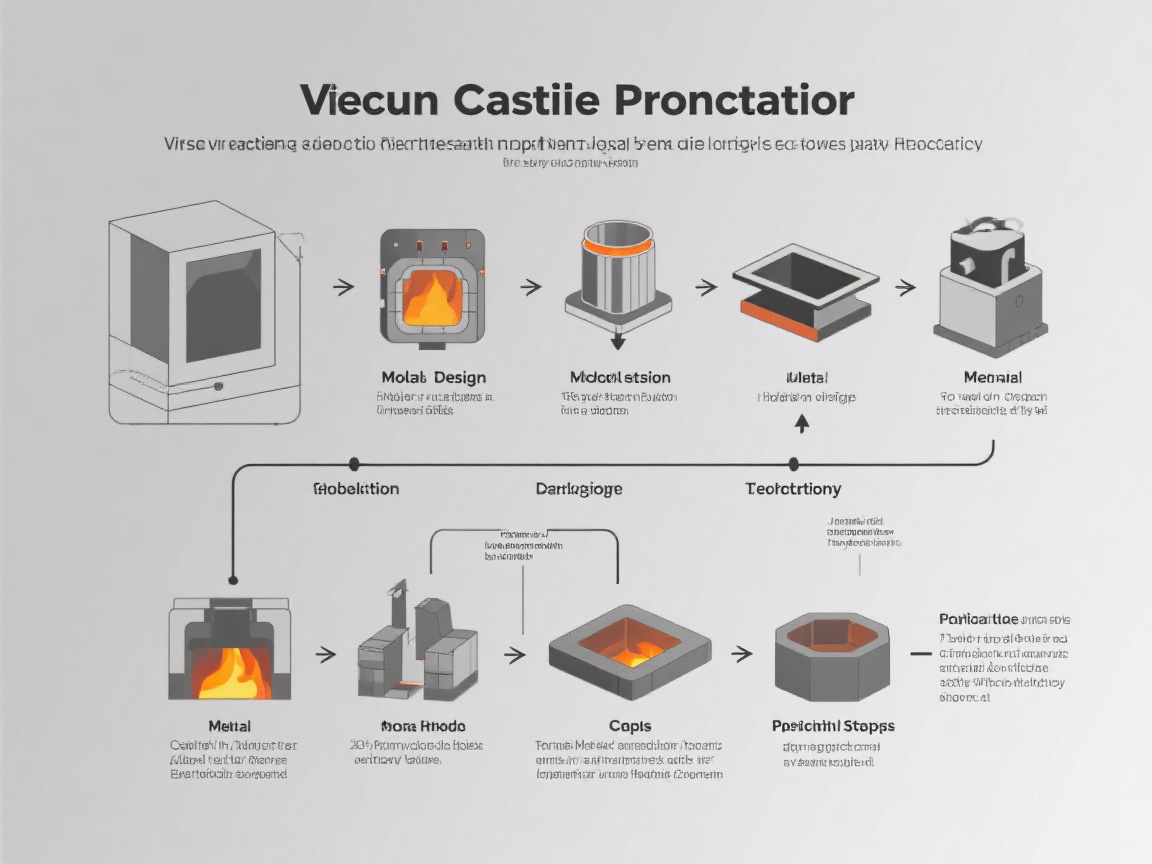
Figure 4: 7-step customization process from initial design to final delivery.
Step-by-Step Customization
- 1
Design Evaluation
Our engineering team reviews your design files to assess feasibility, material suitability, and manufacturing requirements.
- 2
Master Model Creation
Precision CNC machining or 3D printing creates the master model with ±0.02mm tolerance, verified by CMM inspection.
- 3
Silicone Mold Fabrication
Medical-grade silicone mold cured at 60°C for 4-8 hours, precision cut to reveal the cavity.
- 4
Vacuum Casting
Material mixing under vacuum, casting at ≤5 mbar pressure, and controlled curing at 40-80°C.
- 5
Quality Inspection
100% visual inspection, optical comparator for critical dimensions, and material property verification.
- 6
Surface Treatment
Optional finishing processes including painting, coating, sterilization, and packaging.
- 7
Final Delivery
Certified products delivered with complete documentation including material certificates and test reports.
Applications: Where Vacuum Casting Shines in Healthcare

Figure 5: Comprehensive range of medical devices produced through vacuum casting.
Surgical instruments
custom grips, ergonomic handles
Diagnostic devices
probe covers, sensor housings
Patient care
orthopedic braces, prosthetic liners
Drug delivery
infusion pump parts, inhalers
example: custom orthotics for rare conditions, low-volume production with exact fit. Our vacuum casting process allows for patient-specific designs that would be cost-prohibitive with traditional manufacturing methods.
Quality Control & Certifications
Our ISO 13485-certified quality management system ensures every part meets regulatory and performance standards. We implement rigorous testing at every stage of production:
- Dimensional inspection using optical comparators and CMM
- Biocompatibility testing per ISO 10993 standards
- Sterilization validation (autoclave, EtO, gamma radiation)
- Surface finish inspection (Ra ≤0.8μm standard)
- Packaging integrity testing for sterile products
🏆
Certifications
FAQs About Medical Device Vacuum Casting
Q: Max part size?
A: Goldcattle casts up to 300mm x 200mm x 150mm — ideal for handheld devices.
Q: Sterilizable?
A: Yes, resins compatible with autoclave (134°C), EtO, gamma. Critical for reusable tools.
Q: Mold life?
A: Medical silicone molds yield 20–50 parts (high-precision). Rigid resins up to 500 parts.
Q: Clear/translucent parts?
A: Clear PU/silicone with 90% transmittance for IV sets, diagnostic windows.
Q: Lead time for prototypes?
A: Typical 3-5 days for mold creation, 1-2 days for casting. Emergency service available.
Q: Material color options?
A: Full range of FDA-approved pigments, including custom color matching.
Q: Minimum order quantity?
A: As low as 10 parts for prototyping, up to 1000 parts for production runs.
Q: Post-processing options?
A: Painting, coating, texturing, sterilization, and custom packaging.
Ready to Bring Your Medical Device to Life?
Medical device vacuum casting bridges precision, speed, and cost-effectiveness. Goldcattle combines regulatory expertise, advanced technology, and biocompatibility to deliver parts that advance patient care.
📞 +86-18150097490
Working on a prototype or low-volume production? Our vacuum casting specialists are ready to help. Send us your design files for a free feasibility assessment.






