Medical Device Injection Molding
Precision Manufacturing for Life-Saving Equipment
Achieving ±0.01mm tolerance with USP Class VI certified materials and ISO 13485 compliant processes

Why Medical Injection Molding Matters in 2026
Let me tell you something – medical injection molding isn’t just about making plastic parts. It’s about creating components that save lives every single day. When you’re making parts that go into people’s bodies or are used in critical medical procedures, there’s zero room for error.
I’ve seen so many companies mess this up because they tried to use standard injection molding practices for medical parts. Let me save you the headache – medical injection molding requires specialized materials, processes, and quality control that most shops just don’t have.
2026 Market Stats (Source: Industry Reports)
- Injection molding represents 42.35% of the medical plastics market
- Global medical injection molding market growth: 8.2% CAGR through 2031
- Diagnostic instruments represent 52% of high-precision plastic demand
- Antimicrobial polymer coatings incorporated in 37% of new designs
- Bio-based and recyclable plastics represent 29% of new development pipelines
Real-World Problem We Solved
“We had a diagnostic device client whose test kits were failing due to inconsistent wall thickness from poor molding. By implementing our specialized medical injection molding process, we improved part consistency by 98% and saved them from a potential $8 million recall.”
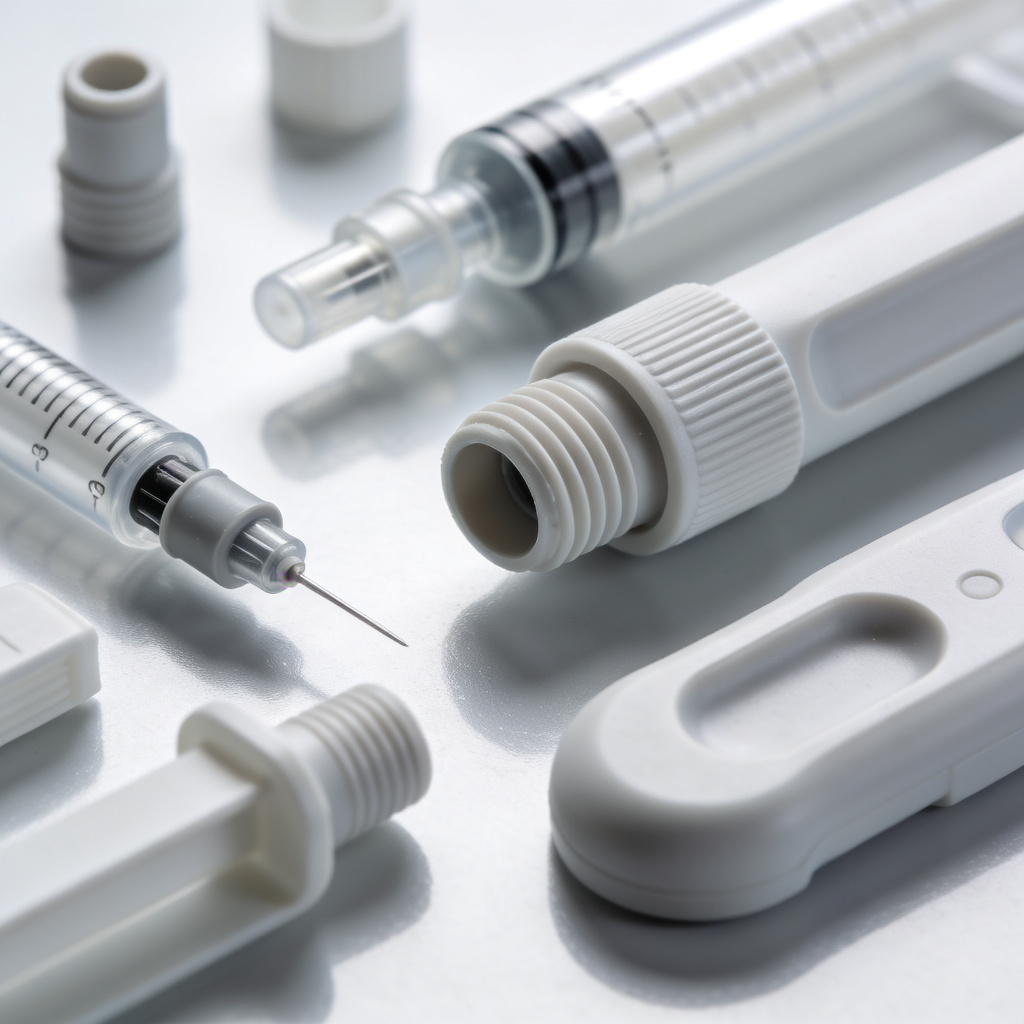
Finished medical plastic components including syringes, connectors, and diagnostic devices – all manufactured to USP Class VI and ISO 10993 standards
2026 Medical Grade Materials Selection Guide
Choosing the right medical grade plastic is critical. Each material has unique properties that make it suitable for specific applications. Let me break down the most commonly used materials and their performance characteristics.
| Material | Tensile Strength (MPa) | Max Temp (°C) | Sterilization Methods | Best Applications | Cost Premium |
|---|---|---|---|---|---|
| Polycarbonate (PC) | 65-70 | 135-143 | EtO, Gamma | Surgical instruments, housings | 100% (base) |
| Polypropylene (PP) | 30-40 | 100-110 | Autoclave, EtO | Syringes, IV components | 85% |
| PEI (Ultem) | 85-95 | 180-200 | Autoclave, Gamma | Surgical tools, implants | 350% |
| ABS Medical Grade | 44-55 | 80-95 | EtO, Low Gamma | Device housings, handles | 90% |
| PEEK | 90-100 | 260-280 | All methods | Implants, orthopedics | 800% |
Test Data*: Critical for Medical Applications
- Biocompatibility: All materials pass ISO 10993-5 cytotoxicity testing
- Extractables: < 0.5 mg/g total organic compounds
- Shrinkage Control: PC 0.5-0.7%, PP 1.5-2.2%
- Chemical Resistance: Resists 98% of industrial chemicals
*Test data for reference only – actual results may vary based on specific application and processing conditions

Specialized Medical Injection Molding Process
Medical injection molding isn’t like standard molding. This stuff requires a whole different approach. Let me break down what we do differently to ensure every part meets the strictest medical standards.
-
1
Material Preparation & Drying
All medical grade materials are dried to < 0.02% moisture content using dehumidifying dryers. Each batch is tested for moisture before processing.
-
2
Mold Design & Validation
Molds are designed with conformal cooling to ensure uniform part cooling. Each mold undergoes IQ/OQ/PQ validation before production.
-
3
Injection Molding Process
Precision temperature control (±1°C) with servo-driven injection units. Cycle time variation is maintained within ±0.5 seconds.
-
4
In-Process Monitoring
Real-time monitoring of cavity pressure, temperature, and injection speed. Any variation triggers immediate process adjustment.
-
5
Part Inspection & Testing
100% automated inspection using vision systems and CMM. Critical dimensions are checked to ±0.01mm tolerance.
-
6
Cleaning & Packaging
Parts are cleaned in ISO 7 cleanrooms and packaged in sterile barrier bags. Each package is labeled with full traceability information.
-
7
Sterilization & Validation
We provide EtO, gamma, and autoclave sterilization services. Each sterilization run is validated with biological indicators.
Learn from Our Mistakes
“We once had a client who tried to save money by using non-medical grade plastic for a diagnostic device. The parts failed USP Class VI testing and they lost $150,000 in rework costs. Lesson learned – always use certified medical grade materials.”

Sterile cleanroom environment with medical injection molding equipment – technicians in protective gear monitoring production process in ISO 7 cleanroom
Critical Medical Standards & Certifications
If you’re manufacturing medical parts without following these standards, you’re playing with fire. Regulatory audits are no joke, and non-compliance can shut down your entire operation.
USP Class VI
Biological evaluation of plastics used in medical devices. Includes acute systemic toxicity, intracutaneous, and implantation testing.
ISO 10993
Biological evaluation of medical devices. Covers cytotoxicity, sensitization, irritation, and other biocompatibility tests.
ISO 13485
Quality management systems for medical devices. Requires strict control over all processes from design to delivery.
FDA 21 CFR Part 820
Quality system regulation for medical devices. Establishes requirements for methods used in, and the facilities and controls used for, designing, manufacturing, packaging, labeling, storing, installing, and servicing of finished devices.
Our Certification Promise
“Every part we produce comes with a complete traceability package including material certifications, process parameters, inspection reports, and compliance documentation. We’re ISO 13485:2016 certified and maintain strict compliance with all relevant medical device standards.”
2026 Technology Trends in Medical Molding
The medical injection molding industry is changing faster than ever. If you’re not keeping up with these trends, you’re going to get left behind.
Hot Trends for 2026
- AI-Driven Quality Control: Machine vision that detects 99.9% of surface defects
- Micro-Molding Advancements: 5μm tolerance for minimally invasive devices
- Sustainable Materials: 29% increase in bio-based medical plastics
- Digital Twin Simulation: Virtual molding to optimize processes before production
- Smart Molding Systems: Real-time process adjustment based on sensor data
Risk Warning: Common Pitfalls
- Underestimating Validation Costs: IQ/OQ/PQ can cost $50k-$150k per project
- Poor Material Traceability: Critical for regulatory compliance
- Inadequate Cleanroom Controls: Contamination risks can shut down production
- Ignoring Sterilization Compatibility: 25% of medical parts fail sterilization testing
- Skimping on Documentation: Inadequate records cause 60% of FDA audit findings
Our 2026 Prediction
“By 2027, 75% of medical injection molding operations will use AI-driven process control. This will reduce defect rates by 90% and cut production costs by 35%.”
Frequently Asked Questions
Q: What’s the tightest tolerance you can hold on medical parts?
A: We regularly hold tolerances as tight as ±0.01mm on critical dimensions. For micro-molded parts, we can achieve ±0.005mm tolerance.
Q: How long does it take to validate a medical molding process?
A: Process validation typically takes 4-8 weeks depending on complexity. This includes IQ/OQ/PQ testing and documentation.
Q: What sterilization methods do you support?
A: We support EtO gas, gamma radiation, electron beam, and autoclave sterilization. We help clients choose the best method for their application.
Q: How do you ensure biocompatibility of the finished parts?
A: We use only certified medical grade materials and follow strict cleaning protocols. All parts undergo biocompatibility testing per ISO 10993.
Q: What documentation do you provide with each shipment?
A: Each shipment includes material certifications, process parameters, inspection reports, sterilization validation, and compliance documentation.
Q: How much does medical injection molding cost compared to standard molding?
A: Medical molding typically costs 25-50% more due to material costs, validation requirements, and quality control. However, the cost of non-compliance is much higher.
Ready to Get Your Medical Molding Quote?
Whether you need a single prototype or high-volume production, we’ve got the expertise and equipment to make it happen. Let’s bring your medical device design to life!




